Author: Dr. KUMARASWAMI SIVAN
What is Cellular Staining?
Cell staining is a technique that can be used to better visualize cells and cell components under a microscope. By using different stains, one can preferentially stain certain cell components, such as a nucleus or a cell wall, or the entire cell. Most stains can be used on fixed, or non-living cells, while only some can be used on living cells; some stains can be used on either living or non-living cells.
Why Stain Cells?
The most basic reason that cells are stained is to enhance visualization of the cell or certain cellular components under a microscope. Cells may also be stained to highlight metabolic processes or to differentiate between live and dead cells in a sample. Cells may also be enumerated by staining cells to determine biomass in an environment of interest.
How Are Cells Stained and Slides Prepared?
Cell staining techniques and preparation depend on the type of stain and analysis used. One or more of the following procedures may be required to prepare a sample:
- Permeabilization – treatment of cells, generally with a mild surfactant, which dissolves cell membranes in order to allow larger dye molecules to enter inside the cell.
- Fixation – serves to “fix” or preserve cell or tissue morphology through the preparation process. This process may involve several steps, but most fixation procedures involve adding a chemical fixative that creates chemical bonds between proteins to increase their rigidity. Common fixatives include formaldehyde, ethanol, methanol, and/or picric acid.
- Mounting – involves attaching samples to a glass microscope slide for observation and analysis. Cells may either be grown directly to the slide or loose cells can be applied to a slide using a sterile technique. Thin sections (slices) of material such as tissue may also be applied to a microscope slide for observation.
- Staining – application of stain to a sample to color cells, tissues, components, or metabolic processes. This process may involve immersing the sample (before or after fixation or mounting) in a dye solution and then rinsing and observing the sample under a microscope. Some dyes require the use of a mordant, which is a chemical compound that reacts with the stain to form an insoluble, colored precipitate. The mordanted stain will remain on/in the sample when excess dye solution is washed away.
What Are Some Common Stains?
There are several types of staining media, each can be used for a different purpose. Commonly used stains and how they work are listed below. All these stains may be used on fixed, or non-living, cells and those that can be used on living cells are noted.
- Bismarck Brown – colors acid mucins, a type of protein, yellow and may be used to stain live cells
- Carmine – colors glycogen, or animal starch, red
- Coomassie blue – stains proteins a brilliant blue, and is often used in gel electrophoresis
- Crystal violet – stains cell walls purple when combined with a mordant. This stain is used in Gram staining
- DAPI – a fluorescent nuclear stain that is excited by ultraviolet light, showing blue fluorescence when bound to DNA. DAPI can be used in living of fixed cells
- Eosin – a counterstain to haematoxylin, this stain colors red blood cells, cytoplasmic material, cell membranes, and extracellular structures pink or red.
- Ethidium bromide – this stain colors unhealthy cells in the final stages of apoptosis, or deliberate cell death, fluorescent red-orange.
- Fuchsin – this stain is used to stain collagen, smooth muscle, or mitochondria.
- Hematoxylin – a nuclear stain that, with a mordant, stains nuclei blue-violet or brown.
- Iodine – used as a starch indicator. When in solution, starch and iodine turn a dark blue color.
- Malachite green – a blue-green counterstain to safranin in Gimenez staining for bacteria. This stain can also be used to stain spores.
- Methylene blue – stains animal cells to make nuclei more visible.
- Neutral/Toluene red – stains nuclei red and may be used on living cells.
- Nile blue – stains nuclei blue and may be used on living cells.
- Osmium tetroxide – used in optical microscopy to stain lipids black.
- Rhodamine – a protein-specific fluorescent stain used in fluorescence microscopy.
- Safranin – a nuclear stain used as a counterstain or to color collagen yellow.
After staining cells and preparing slides, they may be stored in the dark and possibly refrigerated to preserve the stained slide, and then observed with a microscope.
Introduction to Microorganisms – Bacteria
Microorganisms are living organisms that include bacteria, fungi, protozoa, and viruses. They are so small that they require the aid of a microscope to visualize, sometimes even an electron microscope. Bacteria, fungi, and protozoa can be identified and classified with histochemical procedures, and viruses are generally identified with immunohistochemical procedures.
The Gram Stain
Scientists will often choose to perform a differential stain, as this allows them to gather additional information about the bacteria they are working with. Differential stains use more than one stain, and cells will have a different appearance based on their chemical or structural properties. Some examples of differential stains are the Gram stain, acid-fast stain, and endospore stain. You will learn how to prepare bacterial cells for staining, and learn about the gram staining technique.
This very commonly used staining procedure was first developed by the Danish bacteriologist Hans Christian Gram in 1882 (published in 1884) while working with tissue samples from the lungs of patients who had died from pneumonia. Since then, the Gram stain procedure has been widely used by microbiologists everywhere to obtain important information about the bacterial species they are working with. Knowing the Gram reaction of a clinical isolate can help the health care professional make a diagnosis and choose the appropriate antibiotic for treatment.
Gram stain results reflect differences in cell wall composition. Gram positive cells have thick layers of peptidoglycan (a carbohydrate) in their cell walls; Gram negative bacteria have very little. Gram positive bacteria also have teichoic acids, whereas Gram negatives do not. Gram negative cells have an outer membrane that resembles the phospholipid bilayer of the cell membrane. The outer membrane contains lipopolysaccharides (LPS), which are released as endotoxins when Gram negative cells die. This can be of concern to a person with an infection caused by a gram negative organism.
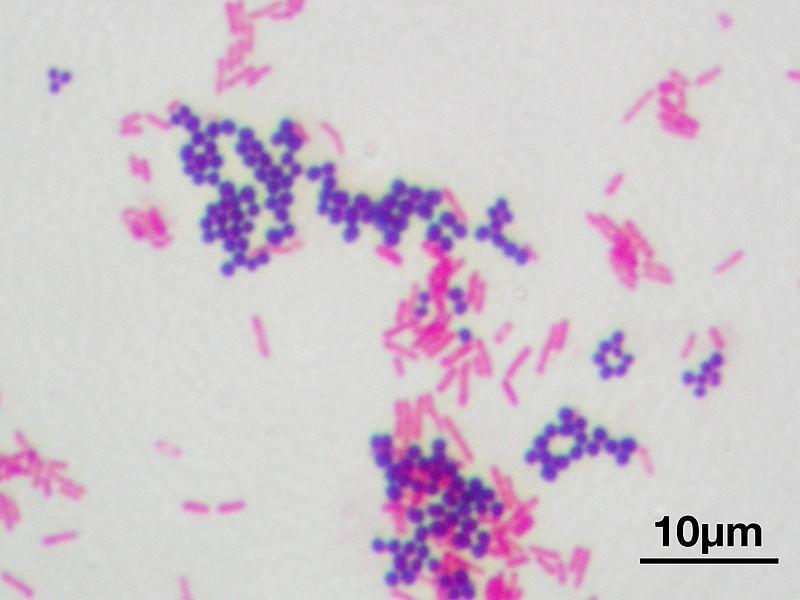
(Staphylococcus aureus ATCC 25923, purple) and Gram-negative bacilli (Escherichia coli ATCC 11775, red). Magnification:1,000. Image by Y Tambe. https://commons.wikimedia.org/wiki/F…m_stain_01.jpg
Mycobacterium tuberculosis, Mycobacterium leprae, and Helicobacter pylori are a few diagnostically relevant special staining methods still relevant in Microscopy
Mycobacterium tuberculosis
It causes tuberculosis, is non-motile and rod-shaped, and is characterized and identified by a property known as “acid-fast” meaning that once absorbed, the dye used to stain the bacilli should not be removed when incubated in an acid rinse. The preferred method of identification is an AFB stain, otherwise called an acid-fast bacteria stain.
Mycolic acid is the mechanism responsible for the acid-fast property. The walls of the bacteria produce this waxy substance called mycolic acid. This acid links with substances within the cell walls of the bacteria, making them resistant to decolorization with acid alcohol treatment. The most common choices of AFB stains are Kinyouin’s carbol fuchsin or Ziehl-Neelsen carbol fuchsin. The solutions, procedures, and results are similar for both.
The Results
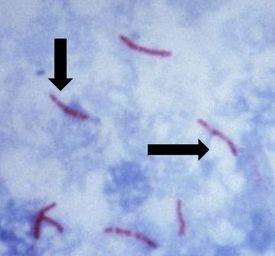
(https://upload.wikimedia.org/wikipedia/commons/0/04/Sputum_smear_for_AFB.jpg)
Mycobacterium leprae
It causes leprosy, has acid-fast properties similar to Mycobacterium tuberculosis but much less effective. The preferred stain for demonstration is Fite’s Faraco.
The Results
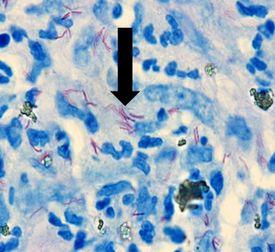
Helicobacter pylori
It is spiral-shaped and is associated with gastric inflammation, peptic ulcers, and gastric carcinoma. They have argyrophilic properties and cannot absorb or reduce silver. The preferred stains for demonstration are Warthin Starry silver or one of many modifications, Giemsa, and immunohistochemistry procedures (most sensitive).
The Results
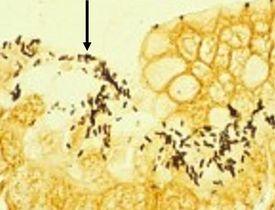
Introduction to Microorganisms – Fungi
Fungi consist of both single and multicellular organisms with distinct nuclei and cell walls. Three common fungi known to cause diseases in humans are Cryptococcus neoformans, Histoplasma capsulatum, and Pneumocystis carinii.
Cryptococcus Neoformans
It lives in the environment throughout the world and can cause meningitis in humans. Infections usually occur in immunocompromised individuals. Capsules are clearly demonstrated by the mucicarmine stain as well as by a methenamine silver procedure.
The Results
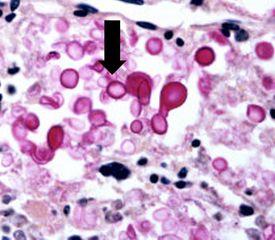
Histoplasma capsulatam
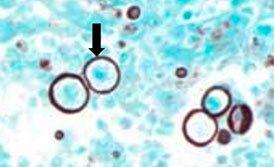
It causes histoplasmosis. Infection occurs from breathing in these spores, which results in flu-like symptoms. If left untreated, it can disseminate throughout the body and cause death. It is more common in immunocompromised patients. It can be demonstrated by the Gomori’s methenamine silver stain.
Pneumocystis carinii
It was previously classified as protozoa but is now considered a fungus-based on nucleic acid and biochemical analysis. It can lead to a fatal pneumonia in immunosuppressed patients. It can be demonstrated by the Gomori’s methenamine silver stain.
Gomori’s methenamine silver
Gomori’s methenamine silver relies on cells that have an argentaffin property. It can reduce invisible silver ions to visible metallic silver without an external aid and is opposite of argyrophilic as seen in Helicobacter pylori
The Results
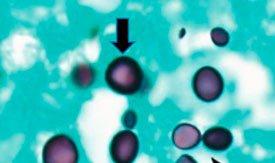
PNEUMOCYSTIS APPEAR AS BLACK BALLS WITH LIGHT GREEN BACKGROUND
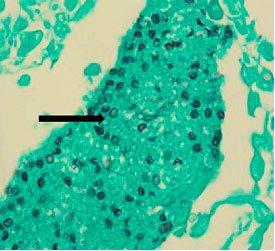
CRYPTOCOCCUS WITH GMS
HISTOPLASMOSIS WITH GMS
Why Digital Microscopy?
A digital microscope is a modern microscope that lacks an eyepiece, an immense difference from the traditional optical microscope. They have a digital camera which is the detector and the image output device. The display of the image is done on a computer screen or monitor, defining the microscope’s digital scope.
This microscope’s light source is an inbuilt LED-source as compared to the optical microscope whose light source is accessed from outside the microscope through an eyepiece. Therefore, in the digital microscope, human optics access is eliminated since the entire apparatus has an image monitoring system.
There are variants of the digital Microscope including the USB digitalized microscopes which are very expensively industrialized digital microscopes, such as the Kohler illumination and the Phase-contrast Illumination, with installed webcams and macro lens.
The first digital Microscope was manufactured in 1986 in Tokyo, Japan which constituted a control box and a lens the was connected to the camera. This is currently known as the Hirox Co. LTD. Because of its computerized connection, it can handle very large digital data obtained from the digitalized camera installed within the microscope. In 2005, a more advanced digital microscope was manufactured which did not require a computer, instead, it had an in-built unit that constituted a monitor and a computer. In 2015, however, a new digitalized microscope was also constructed, with an external computer, that has a USB connection, which expanded on the longevity and speed of the computer. This reduced the external cable connections along with the size of the computer itself was reduced.
They have an image processing software, that adjusts the image brightness, enhances the contrast, scale, and crop the image.
The development of the digital Microscope takes into account two of its major functions. The Input and out functions and in this aspect, the digital microscope is made of two major parts;
- The hardware – this is majorly the analog part of the microscope, with a light source, the analog microscope, a camera, and the camera components. the Camera replaces the eyepiece of the traditional microscope. The image from the specimen gets focused on the camera and it is displayed on the computer monitor screen which is then stored and can also be processed.
- The software – This is the section of the microscope that has the camera drivers and the image processing software, which has organized units which include the viewer unit, brightness adjustment unit, image contrast unit, histogram equalization unit, image scaling unit and the image cropping unit. The image viewer unit continuously displays the specimen and captures the image from the microscope which is stored and processed depending on the user’s preferences. The image brightness adjustment unit enhances the brightness of the image depending on how much light is focused on the specimen. It controls the intensity of the light in pixels.
Optical microscopy is a well-established tool in biological research. In modern microscopy, analogue light detectors like the human eye or photographic film are replaced with a digital camera. Digital microscopy comprises image formation by optics, image registration by a digital camera, and saving of mage data in a computer file. Making optimal use of digital microscopy requires taking into account limitations that are particular to each of these processes.
Digital microscopes are considered a relatively new type of inspection and measurement technology. The image collected by the camera is shown in real-time on a built-in display. Their ability to cover a vast magnification range makes them valuable tools for the diagnostic world in Anatomical pathology, Cytopathology, Microbiology, R&D activities across many fields, quality and failure analysis and manufacturing-based applications.
Advantages of Digital Microscopes
The key advantage being as follows:
- Observe, measure and record with a single system in real-time
- Provide a continuous and wide magnification of images
- Make key observations without the need for cumbersome eyepieces.
- They provide high-resolution magnification of the images in pixels.
- It can tilt and provide a 2D and 3D image measurements.
- They can store huge amounts of data, through imaging and recording movable and unmovable specimens
- They are one-stop operation machines since the visualize the images and provide an output of the images through the computer monitors.
CILIKA has combined technology to be user friendly and also look at ease to exchange views and thoughts thus combining education and diagnostic practice
The Cilika portable series microscope is the perfect product for those who require high quality imaging on the go. From work to home, from lab to field or from practicals to seminars, the Cilika portable microscopes can travel with you while For microscope users who prefer the familiarity and sturdiness of traditional desktop microscope designs, but desire the advantages of a digital microscope, Cilika Benchtop is the way to go.
Experience the boon of microscopy, user friendly technology and the right solutions and mix of optical and the digital world for Histopathologists, Cytologists. Microbiologists, Mycologists and Haematologist
